Revolutionizing H2O2 Production: Efficient Electrochemical Synthesis with Single-Atom Catalysts
Key Ideas
- Introduction of efficient electrochemical method for H2O2 production using single-atom catalysts shows promising results.
- The Ni-SAC electrode demonstrates high Faradaic efficiency and H2O2 yield rate, surpassing previous works in the field.
- The cooperative mechanism of Ni atom for O2 adsorption and carbon nanosheets for H* generation significantly enhances H2O2 production.
- The coupling system of 2e− ORR and ethylene glycol oxidation reaction not only produces H2O2 and glycolic acid simultaneously but also shows economic viability with a higher profit margin.
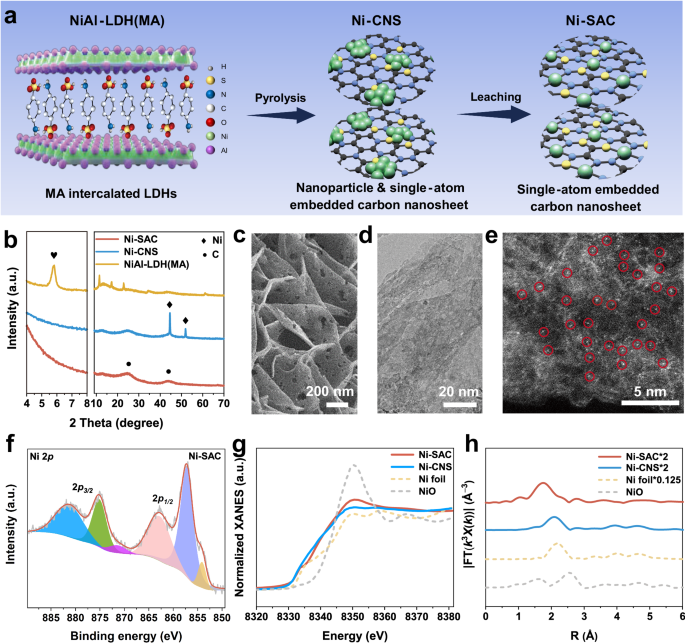
Hydrogen peroxide (H2O2) is a vital chemical used in various industries globally. The traditional anthraquinone cycling method for its production has drawbacks of organic waste generation and high transport costs. A more efficient and environmentally friendly production method is needed. The article introduces an innovative approach utilizing electrochemical synthesis via single-atom catalysts (SACs). The process aims at increasing H2O2 yield through the 2e− ORR pathway. An electrode containing a single-atom Ni catalyst (Ni-SAC) exhibited exceptional performance with high Faradaic efficiency and H2O2 yield rate, outperforming existing methods. The cooperative mechanism of O2 adsorption by the Ni atom and H* generation by carbon nanosheets significantly enhanced H2O2 production. Moreover, a coupling system combining the 2e− ORR at the cathode and ethylene glycol oxidation at the anode allowed simultaneous production of H2O2 and glycolic acid, demonstrating economic viability. The article concludes with a techno-economic evaluation showing a higher profit margin for the coupling system, indicating promising future applications.
Topics
Production
Technology
Innovation
Sustainability
Energy Efficiency
Manufacturing
Research
Nanotechnology
Chemistry
Latest News